Summary
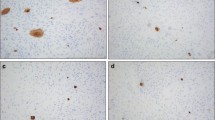
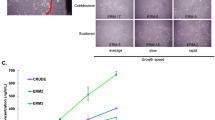
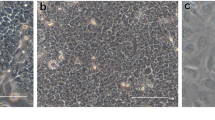
To study biological character and function of epithelial rests of Malassez (ERM) in human periodontal ligament, we have developed a serum-free culture system of epithelial cells (ME) derived from ERM. The mitogenic effects of fibroblast growth factor (FGF)-1, FGF-2, and FGF-7/keratinocyte growth factor (KGF) on ME, human periodontal ligament-derived fibroblasts (PLF), human oral epithelial cells (OE), and human submandibular gland-derived epithelial cells (SGE) were investigated under a serum-free culture condition. FGF-1 and FGF-7/KGF stimulated the growth of both ME and SGE but FGF-2 had no effect. On the other hand, FGF-1, FGF-2, and FGF-7/KGF increased the OE proliferation. These results suggested that the divergent requirement of FGF ligands among these cells would be attributed to the different expression pattern of FGF receptor (FGFR) messenger ribonucleic acid (mRNA) isotypes. Therefore, we examined the expression of FGFR isotypes in these cells. Reverse transcription-polymerase chain reaction (RT-PCR) analysis of ME- and SGE-derived mRNAs revealed the presence of mRNA encoding FGFR2-IIIb, a high affinity receptor for FGF-1 and FGF-7/KGF. However, no mRNAs for other FGFR isotypes were detected in both ME and SGE. On the contrary, OE expressed FGFR1-IIIc, FGFR3-IIIb, and FGFR4 mRNAs as well as FGFR2-IIIb. These results indicate that FGF binding sites on ME dominantly bind to FGF-1 and FGF-7/KGF, which transduce their signals via FGFR2-IIIb. Immunohistochemical analysis, PCR-Southern, ribonuclease protection assay (RPA), and Western blotting revealed that PLF expressed FGF-7/KGF mRNA and its peptide. These observations suggest that FGF-7/KGF might mediate epithelial-mesenchymal interactions between ME and PLF to maintain normal structure and function of periodontal ligament.
Similar content being viewed by others
References
Aviv, H.; Leder, P. Purification of biologically active globulin messenger RNA by chromatography on oligothymidylic acid cellulose. Proc. Natl. Acad. Sci. USA 69:1408–1412; 1972.
Burgess, W. H.; Maciag, T. The heparin-binding (fibroblast) growth factor family of proteins. Ann. Rev. Biochem. 58:575–606; 1989.
Chellaiah, A. T.; McEwen, D. G.; Werner, S., et al. Fibroblast growth factor receptor (FGFR)-3. Alternative splicing in immunoglobulin-like domain III creates a receptor highly specific for acidic FGF/FGF-1. J. Biol. Chem. 269:11,620–11,627; 1994.
Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162:156–159; 1987.
Emoto, H.; Tagashira, S.; Mattei, M. G., et al. Structure and expression of human fibroblast growth factor-10. J. Biol. Chem., 272:23,191–23,194; 1997.
Finch, P. W.; Rubin, J. S.; Miki, T., et al. Human KGF is FGF-related with properties of a paracrine effector of epithelial cell growth. Science 245:752–755; 1989.
Furue, M.; Okamoto, T.; Ikeda, M., et al. Primitive neuroectodermal tumor cell lines derived from a metastatic pediatric tumor. In Vitro Cell. Dev. Biol. 30A:813–816; 1994.
Gao, Z.; Flaitz, C. M.; Mackenzie, I. C. Expression of keratinocyte growth factor in periapical lesions. J. Dent. Res., 75:1658–1663; 1996.
Hou, J.; Kan, M.; McKeehan, K., et al. Fibroblast growth factor receptors from liver vary in three structural domains. Science 251:665–668; 1991.
Jaye, M.; Schlessinger, J.; Dionne, C. A. Fibroblast growth factor receptor tyrosine kinases: molecular analysis and signal transduction. Biochem. Biophys. Acta 1135:185–199; 1992.
Johnson, D. E.; Lu, J.; Chen, H., et al. The human fibroblast growth factor receptor genes: a common structural arrangement underlies the mechanisms for generating receptor forms that differ in their third immunoglobulin domain. Mol. Cell. Biol., 11:4627–4634; 1991.
Kawasaki, E. S.; Clark, S. S.; Coyne, M. Y., et al. Diagnosis of chronic myeloid and acute lymphocytic leukemias by detection of leukemia-specific mRNA sequences amplified in vitro. Proc. Natl. Acad. Sci. USA 85:5698–5702; 1988.
Klagsbrun, M. The fibroblast growth factor family: structural and biological properties. Prog. Growth Factor Res. 1:207–235; 1989.
Kobrin, M. S.; Yamanaka, Y.; Friess, H., et al. Aberrent expression of type I fibroblast growth factor receptor in human pancreatic adenocarcinomas. Cancer Res., 53:4741–4744; 1993.
Kvam, E.; Gilhuus-Moe, O. Uptake of 3H-thymidine by an epithelial rest in the periodontal membrane. A preliminary report. Acta Odont. Scand. 28:143–146; 1970.
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685; 1970.
Lindskog, S.; Blomlof, I.; Hammarstrom, L. Maintaining the periodontal space. J. Clin. Periodontol. 15:371–373; 1988.
Loe, H.; Waerhaug, J. Experimental replantation of teeth in dogs and monkeys. Arch. Oral Biol. 3:176–184; 1961.
Main, D. M. G. Odontogenic epithelial residues around rabbit mandibular cheek teeth [abstract #7]. J. Dent. Res. 51:841; 1972.
Malassez, L. Sur l'existence de masses epithelailes dans le ligament alveolodentaire chez l'homme adulte et a l'etat normal. Comptes Rendus Societe de Biologie 36:241–244; 1884.
Mason, I. J. The ins and outs of fibroblast growth factors. Cell 78:547–552; 1994.
Miki, T.; Fleming, T. P.; Bottaro, D. P., et al. expression of cDNA cloning of the FGF receptor by creation of a transforming autocrine loop. Science 251:72–75; 1991.
Miki, T.; Bottaro, D. P.; Fleming, C. L. Determination of ligand-binding specificity by alternative splicing: two distinct growth factor receptors encoded by a single gene. Proc. Natl. Acad. Sci. USA 89:246–250; 1992.
Myoken, Y.; Myoken, Y.; Okamoto, T., et al. Immunocytochemical localization of fibroblast growth factor-1 and FGF-2 in oral squamous cell carcinoma. J. Oral. Pathol. Med. 23:451–456; 1994.
Myoken, Y.; Myoken, Y.; Okamoto, T., et al. Effect of fibroblast growth factor—1 on the three dimensional growth and morphogenesis of human salivary gland epithelial cells embedded in collagen gels. In Vitro Cell. Dev. Biol. 31:84–86; 1995.
Okamoto, T.; Yatsuzuka, N.; Tanaka, Y., et al. Growth and differentiation of periodontal ligament-derived cells in serum-free defined culture. In Vitro Cell. Dev. Biol. 33A:302–309; 1997.
Orban, B. The epithelial network in the periodontal membrane. J. Am. Dent. Assoc. 44:632–635; 1952.
Ornitz, D. M.; Xu, J.; Colvin, J. S., et al. Receptor specificity of the fibroblast growth factor family. J. Biol. Chem. 21 271 (25):15,292–15,297; 1996.
Parrott, J. A.; Vigne, J. L.; Chu, B. Z., et al. Mesenchymal-epithelial interactions in ovarian follicle involve keratinocyte and hepatocyte growth factor production by the cells and their action on granulosa cells. Endocrinology 135:569–575; 1994.
Rubin, J. S.; Osada, H.; Finch, P. W., et al. Purification and characterization of a newly identified growth factor specific for epithelial cells. Proc. Natl. Acad. Sci. USA 86:802–806; 1989.
Sanger, F. Determining of nucleotide sequences in DNA. Science 214:1205–1210; 1981.
Sato, J. D.; Kawamoto, T.; Okamoto, T. Cholesterol requirement of P3-X63-Ag8 and X63-Ag8. 653 Mouse myeloma cells for growth in vitro. J. Exp. Med. 165:1761–1766; 1987.
Schendorn, E. T.; Mierindort, R. C. A novel transcription property of SP6 and T7 RNA polymerase: dependence on template and synthetic DNA template. Nucleic Acids Res. 13:6223–6236; 1985.
Shear, M.; Pindborg, J. Microscopic features of the lateral periodontal cyst. Scand. J. Dent. Res. 83:103–110; 1975.
Southern, E. M. Rapid transfer of DNA from agarose gels to nylon membranes. J. Mol. Biol. 98:503–507; 1975.
Spouge, J. D. A study of epithelial odontogenic residues in the pig. J. Periodontol. 57:1164–1171; 1986.
Ten Cate, A. R. The epithelial rests of Malassez and the genesis of the dental cyst. Oral Surg. 34:956–964; 1972.
Yamasaki, M.; Miyake, A.; Tagashira, S., et al. Structure and expression of the rat encoding a novel member of the fibroblast growth factor family. J. Biol. Chem. 5 271 (27):15,918–15,921; 1996.
Yan, G.; Fukabori, Y.; McBride, G., et al. Exon switching and activation of stromal and embryonic fibroblast growth factor (FGF)-FGF receptor genes in prostate epithelial cells accompany stromal independence and malignancy. Mol. Cell. Biol. 13:4513–4522; 1993.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yamanaka, T., Sakamoto, A., Tanaka, Y. et al. Isolation and serum-free culture of epithelial cells derived from epithelial rests of Malassez in human periodontal ligament. In Vitro Cell.Dev.Biol.-Animal 36, 548–553 (2000). https://doi.org/10.1290/1071-2690(2000)036<0548:IASFCO>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2000)036<0548:IASFCO>2.0.CO;2